Reaction Rates
|
Reaction rates vary between different reactions. How reaction rates are measured is by how long reactants take to disappear or how long it takes for a product to appear. According to the collision theory, a product is formed when existing bonds are broken and new bonds are formed.
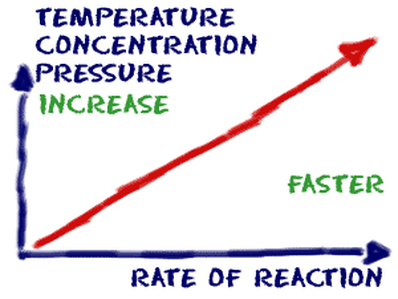
The rate a reaction occurs is affected by different factors such as temperature, concentration, surface area, and the presence of a catalyst. If reactants don’t collide with enough energy, reactions cannot occur. In addition to having enough energy, the collision of reactants must be frequent. The molecules also must be situated correctly to react, which is also known as collision geometry. |
|
First, temperature affects how often and with how much energy the particles in a reaction collide. By increasing temperature, particles will collide more frequently and with more energy.
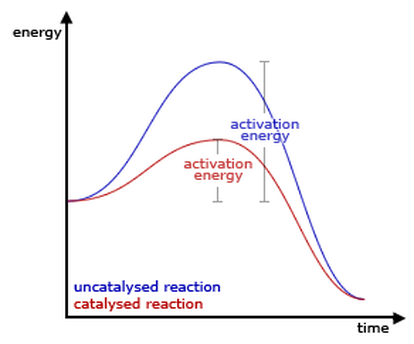
Another way particle collisions can happen more often is increasing the concentration of particles. The more particles there are, the more they are going to hit each other. Atoms and molecules are only able to react at the surface of an object. Increasing the surface area of an object gives atoms and molecules more space to collide, meaning they will collide faster. Catalysts decrease the amount a time a reaction takes to occur by lowering activation energy. To do this, the catalyst finds another way the reaction can occur. Catalysts do not change the heat of a reaction and are able to speed up the a reaction's reverse as well. |
Terms to Know
|
reaction rate:
time it takes for reactants to disappear or time it takes for products to appear collision theory: states that 1. in order to react, molecules must hit each other 2. products are formed by bonds that break then re-bond to form a new bond collision frequency: how often particles hit each other affected by concentration and temperature collision energy: in order for a reaction to happen, this must be present so that when the molecules hit each other, new bonds can be made affected by temperature collision geometry: in order for a reaction to occur, the molecules must be set in a particular way |
catalyst:
speeds up reactions (forward and reverse) by lowering activation energy doesn't change heat of a reaction not consumed by the reaction temperature: increase of this factor leads to increase of collision frequency and energy heat: can be product or reactant increase of shifts equilibrium to left, decrease to the right concentration: increase of this leads to increased collision frequency increase of shifts equilibrium to the products side pressure: only affects gases created when particles collide with container or each other increase of shifts equilibrium to the side with less moles of gas -will not shift if moles are equal surface area: particles will collide faster if there is more space for the atoms and molecules to react |
Citations:
"Rate of Reaction." Chem4Kids.com: Reactions: Rates of Reaction. N.p., n.d. Web. 27 May 2014.
"Equilibrium Lesson 1 Reaction Rates." YouTube. YouTube, n.d. Web. 29 May 2014.
"Info:Main Page." - New World Encyclopedia. N.p., n.d. Web. 29 May 2014.
"Rate of Reaction." Chem4Kids.com: Reactions: Rates of Reaction. N.p., n.d. Web. 27 May 2014.
" Unit 12: Reactions Rates & Equilibrium." Ms. Coole. N.p., n.d. Web. 29 May 2014
"Rate of Reaction." Chem4Kids.com: Reactions: Rates of Reaction. N.p., n.d. Web. 27 May 2014.
"Equilibrium Lesson 1 Reaction Rates." YouTube. YouTube, n.d. Web. 29 May 2014.
"Info:Main Page." - New World Encyclopedia. N.p., n.d. Web. 29 May 2014.
"Rate of Reaction." Chem4Kids.com: Reactions: Rates of Reaction. N.p., n.d. Web. 27 May 2014.
" Unit 12: Reactions Rates & Equilibrium." Ms. Coole. N.p., n.d. Web. 29 May 2014