Equilibrium
|
Equilibrium
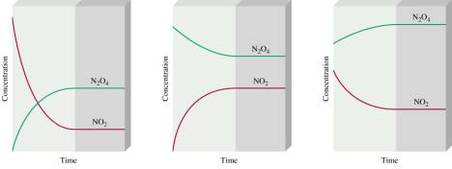
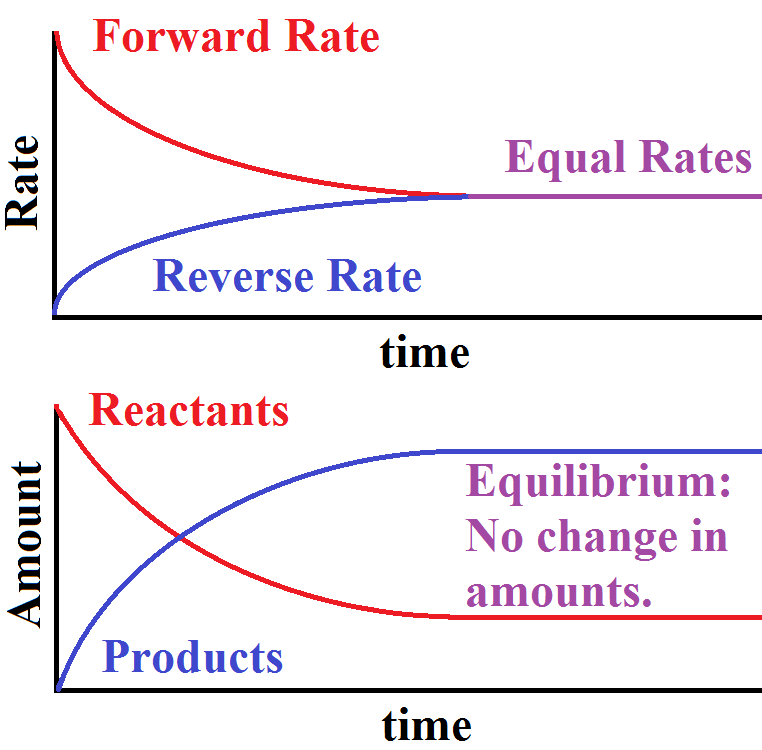
Equilibrium is when the rate of the forward reaction is equal to the rate of a reverse reaction. An equilibrium is a reversible reaction, which means the reaction can go either way. Sometimes the reaction doesn't complete because not all the reactants are used up. The reaction can shift depending on the conditions of the reactants or products. Le Chatelier's Principle
Factors of Le Chatelier's Principle:
When the concentration increases for reactants, the equilibrium moves to the right of the product side. When the concentration increases for the products, the equilibrium moves to the left of the reactant side. Temperature When it comes to heat, it is not a product or reactant. The equilibrium moves to the left when the temperature in increased, and when the temperature is decreased, the equilibrium moves to the right. Pressure The pressure will only affect gases. Two factors pressure cannot affect are liquids and solids. The pressure will shift to the side with the fewest number of moles of gas when increasing. When the moles of gas are equal, there are no shift. When you increase the pressure the equilibrium will got to the right. When you decrease it shifts to the left. |
Citations:
"Chemistry @ Queen's." Chemistry @ Queen's. N.p., n.d. Web. 29 May 2014.
"Equilibrium: Crash Course Chemistry #28." YouTube. YouTube, n.d. Web. 29 May 2014.
"I Love Chem." I Love Chem. N.p., n.d. Web. 29 May 2014.
"Unit 12: Reactions Rates & Equilibrium." Ms. Cool. N.p., n.d. Web. 29 May 2014
"Chemistry @ Queen's." Chemistry @ Queen's. N.p., n.d. Web. 29 May 2014.
"Equilibrium: Crash Course Chemistry #28." YouTube. YouTube, n.d. Web. 29 May 2014.
"I Love Chem." I Love Chem. N.p., n.d. Web. 29 May 2014.
"Unit 12: Reactions Rates & Equilibrium." Ms. Cool. N.p., n.d. Web. 29 May 2014